The project has been closed*.
▼ Updates ▼
2022/6/24 Added event notice and report.

WE HAVE DECIDED TO CHALLENGE CROWDFUNDING IN ORDER TO PROMOTE RESEARCH ACTIVITIES TO MAKE THIS TREATMENT AVAILABLE TO ALL CHILDREN DIAGNOSED WITH ASD AS QUICKLY AND SAFELY AS POSSIBLE.

At present, the cost of transplantation is a significant burden due to the free treatment, and it is not easy to receive intestinal flora transplantation.
The cost of transplantation is high due to fixed costs such as the cost of testing intestinal flora balance using next-generation sequencers, maintenance of donor specimens (JapanBiome), production costs of NanoGAS® water used in the bacterial solution for transplantation (plant maintenance costs, etc.), and personnel costs for managing the research group. The current situation is that the cost of transplantation has become expensive.
Not only in the clinical trials currently taking place at our affiliated medical institutions, but also through clinical research conducted at universities, with the goal of advancing to clinical trials and being recognized as medical care!!!
For those of you who have not had the opportunity to learn about intestinal flora transplantation, we are starting a crowdfunding campaign in the hopes that many people will receive a transplant, that this treatment will spread more widely, and that we can further our research!
We look forward to your warm support. Thank you in advance for your support.
Project Summary
Period] Saturday, May 14 – Sunday, July 10, 2022
Target Amount] 3,000,000 yen
[Purpose of use]
● Various testing costs ● Development costs of automatic purification equipment for bacterial solution ● Gazefinder trial session costs
● Maintenance costs for each plant ● Crowdfunding fees
<Project Implemenation and Period>
Your donation will be used to cover the cost of research to be conducted by the end of fiscal year 2022 (March 31, 2023), which is the end of this project.
Click here to support us (project page)
For return details and support, please click here to preview the page.
We appreciate your cooperation.
Crowdfunding Support Message
From a Patient (Message of support)

I have seen children who have undergone intestinal flora transplantation grow up to be very successful. If I ever meet a child with autism spectrum disorder in the future, I will tell him or her that this is a very good treatment method for the child. I hope that this revolutionary intestinal flora transplantation will spread rapidly.

If intestinal flora transplants become available through insurance, many people will seek treatment. This is because there are so many possibilities for intestinal flora transplantation, including its application for cancer. However, I have heard that we need to collect more cases to bring transplantation to that stage. I know the road will be hard, but we need to increase the number of cases and bring the cost closer to the point where it is affordable. At the same time, research should be further developed. In this way, I hope that the number of people who want to undergo transplantation will increase, even if only by one person.
From Dr. Masahiko Johya, Director, Luke’s Ashiya Clinic (Video)
From Naomi Hatanaka, whose son experienced a transplant (Video)
event preview
July 5, 2022, “Second Annual Loose Fungi and Roundtable Discussion”

We are pleased to welcome Ms. Ai Kitamura, a fermented food specialist, to our online event with the theme of “A diet that makes your intestinal bacteria happy”.
Fermented foods that are good for you?
You mean yogurt or natto?
What are some easy cooking tips?
And so on… Whether you just want to listen or want to talk with us, you’re welcome…
Click the banner above for more information!
Event Report
June 15, 2022 “First Roundtable Discussion with Loose Fungi”
We held a “Roundtable Discussion with Loose Bacteria” online on Wednesday, June 15 at 12:00 p.m.
More than 40 people attended and sent us positive feedback that it was good to hear about the experiences of parents who have actually had transplants and that it has become an institution for learning about intestinal bacteria.
Some of the day’s videos are now available! Please click here to view.
May 14, 2022 Kick-off event
The kick-off event was held as follows.
We have received immediate support and would like to thank everyone for making this kick-off event a success.
We are planning to hold another event at the end of the crowdfunding period, so we hope you will join us.
▼ Kickoff Event
Date & Time] Saturday, May 14, 2022, 11:30 a.m. Opening
Participation fee] Free of charge
Place] Kobe Health Industry Development Center (Hidec) 2F Multipurpose Room (tentative)
6-7-4 Minatojima-Minami-cho, Chuo-ku, Kobe, Hyogo 650-0047, Japan
Kobe Health Industry Development Center (Hidec)
You can also participate online.
Contents】Trial use of the Gazefinder eye tracking device,
Mini seminar on ASD and intestinal bacteria, Roundtable discussion



HELD ONLINE AND AT THE VENUE, THE “UNDERSTANDING ASD AND INTESTINAL BACTERIA MINI-SEMINAR” FIELDED A VARIETY OF QUESTIONS.

Gazefinder is a device that objectively evaluates the characteristics of children with ASD by measuring eye movement. Since it only requires the child to watch a video projected on a monitor screen, even small children can be measured, and it is used in research on ASD and other disorders.

The study group uses Gazefinder to quantitatively evaluate changes before and after transplantation.
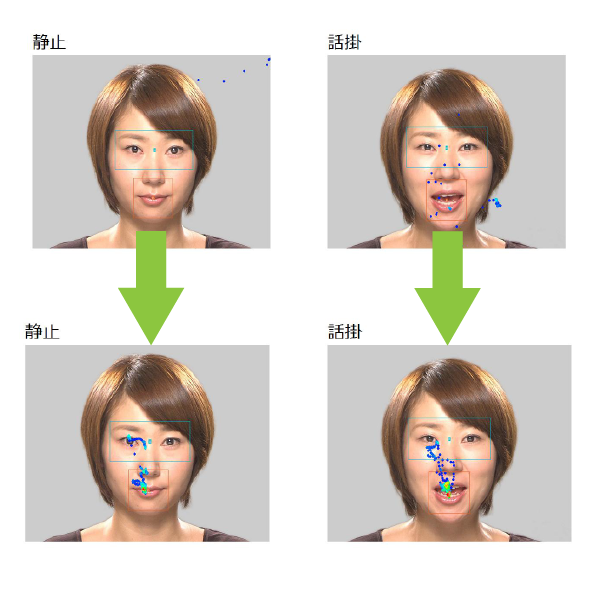
~Gazefinder measurement data before and after intestinal flora transplantation (FMT) ~
The viewpoints that could be measured appear as blue dots.
After transplantation (lower portion), the patient is able to see people’s eyes and mouth clearly.
ASD diagnostic criteria include difficulty with eye contact interactions and discrimination of gaze direction.
In the data below, before the transplant (top), the patient barely looked at the eyes, but after the transplant (bottom), there is a change in gaze direction, looking at the eyes and looking at the mouth.
This is a significant change for people with ASD, who often say that they feel discomfort and stress when making eye contact with others.
The Australian clinical trial for ASD evaluation using the Gazefinder eye tracking device has been completed, and an application for medical device approval has been filed with the Australian Medical Products Administration (TGA).
In addition, an application was filed with the Australian Medical Products Administration (TGA*4) for medical device approval for an ASD evaluation device based on the Gazefinder’s eye measurement technology.
1: University based in Melbourne, Victoria, Australia*2: Medical research institute based in Perth, Western Australia, Australia*3: Autism Spectrum Disorder*4: Australian Therapeutic Goods Administration Administration
Source: JVC KENWOOD Corporation https://www.jvckenwood.com/jp/press/2021/press-211011-01.html
https://www.jvckenwood.com/jp/press/2021/press-211011-01.html
Please click here to support us!
プロジェクト詳細・ご支援はこちら









![Event] Roundtable Discussion with Loose Fungi – A Gathering of Mothers Working Hard for Their Children’s Future SP! ~](https://fmt.sym-biosis.co.jp/wp-content/uploads/2025/02/腸内フローラ移植臨床研究会 学術大会-480x320.png)







