Autism Spectrum Disorder (ASD) is a developmental disorder characterized by challenges in social interaction and communication, often accompanied by a strong preoccupation with specific interests or routines.
Symptoms vary widely among individuals, with significant differences in intellectual development, language ability, and sensory sensitivities. As a result, even with the same “ASD” diagnosis, the specific challenges faced are unique to each person.
Diagnosis is primarily conducted through clinical behavioral observations and psychological testing. Diagnostic criteria for ASD include the DSM-5 (Diagnostic and Statistical Manual of Mental Disorders by the American Psychiatric Association) and ICD-10/11 (International Classification of Diseases by the World Health Organization).
In recent years, there has been a rise in cases of ASD without intellectual disability (so-called high-functioning autism) and individuals who are only diagnosed after reaching adulthood.
ASD is understood to be present from birth as a characteristic of neurodevelopment; it is not something that changes solely through an individual’s efforts or the home environment. Consequently, the goal of support is not to “cure” the condition, but to empower the individual to live a fulfilling life in their own way.
In addition to educational and psychological support, attention has recently shifted toward physical health, particularly the relationship with the intestinal environment. ([Link: https://fmt.sym-biosis.co.jp/en/blog])
What is the Relationship Between Autism Spectrum Disorder (ASD) and the Intestinal Microbiota?
The gut is often referred to as the “second brain,” as the gut and brain are closely connected through nerves, hormones, and the immune system. This interconnectedness is known as the “gut-brain axis.”
Recent research has revealed that the intestinal microbiota (intestinal flora) living in the gut plays a pivotalrole in this connection, leading experts to describe it more accurately as the “microbiota-gut-brain axis.”
It has long been observed that many individuals with ASD experience gastrointestinal symptoms such as constipation and diarrhea. In recent years, reports have shown that beyond these symptoms, the diversity of the intestinal microbiota itself tends to be low in those with ASD.
It has been suggested that disturbances in the intestinal environment may affect neurotransmission and immune responses, potentially contributing to changes in behavior and emotions.
Overseas studies have reported that fecal microbiota transplantation (FMT) may lead to improvements in ASD symptoms. For example, a study at the University of Arizona conducted FMT on 18 patients with ASD and reported behavioral improvements in approximately 40%. Additionally, a study at the Third Military Medical University in China reported roughly 60% improvement four weeks after FMT.
While these studies are still in the exploratory stage, they suggest the potential for maintaining a healthy intestinal environment to influence behavior and emotions.
With NanoGAS®-FMT, we are trialing treatments that gently approach the function of this microbiota-gut-brain axis through the restructuring of the intestinal environment. Detailed features and clinical results are introduced in the following chapter.
Clinical Results and Future Outlook of NanoGAS®-FMT for ASD
Clinical Approach of NanoGAS®-FMT for ASD
NanoGAS®-FMT is a fecal microbiota transplantation method aimed at safely restoring the diversity of intestinal bacteria. ([Link: https://fmt.sym-biosis.co.jp/about])
We have developed this technique to be less invasive than traditional FMT, balancing the intestinal environment in a more natural way.
In Japan, several medical institutions affiliated with The Association for Clinical Research of Fecal Microbiota Transplantation Japan are conducting clinical initiatives for children with ASD. Data from treatment results to date suggest that NanoGAS®-FMT may foster behavioral and emotional changes in individuals with ASD.
Furthermore, beyond simply “improving” symptoms, families have reported transformative changes such as an “increased ease of daily living” and “smoother interactions with family members.”
Evaluations and Tests to Quantify Changes
In NanoGAS®-FMT treatment for ASD, we strive to objectively quantify changes in intestinal flora and behavior before and after transplantation. For children of junior high school age and younger, we combine the following evaluations:
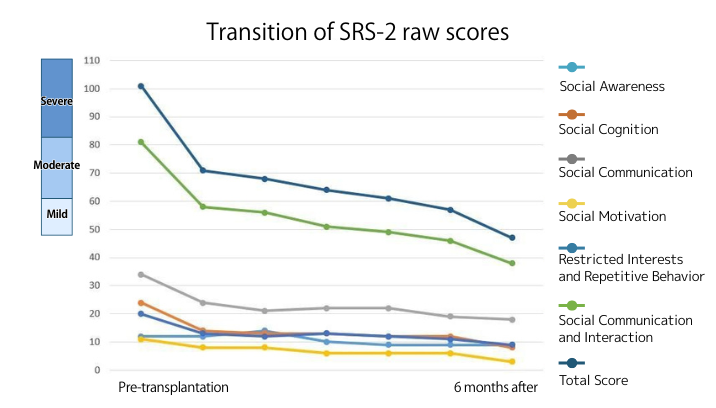
What is SRS-2?
The SRS-2 (Social Responsiveness Scale) is a test that evaluates interpersonal behavior, communication, and repetitive/emotional behaviors—key characteristics of ASD—for individuals ranging from 2.5 years old to adults. It is completed by someone familiar with the subject’s daily life, such as a parent or teacher, and then scored by an examiner.

(Image courtesy of JVCKENWOOD Corporation)
What is “Gazefinder,” the Eye-Tracking Device?
“Gazefinder” is a device that objectively evaluates the characteristics of children with ASD by measuring eye movements. Since it only requires watching a video on a monitor, even young children can participate easily. It is widely used in research on ASD and other conditions.
The Association uses Gazefinder to quantitatively evaluate changes before and after transplantation. By accumulating this data, we continually improve the precision of our future treatments.
Changes in Eye-Tracking Data via Gazefinder
The image below shows eye-tracking data measured before and after fecal microbiota transplantation(NanoGAS®-FMT). The blue dots indicate the subject’s focal points; the top row is before transplantation, and the bottom row is after.
Before transplantation (top row), the gaze is scattered away from the person’s face, failing to properlycapture facial elements such as the eyes and mouth. Conversely, after transplantation (bottom row), the gaze is concentrated on the eyes and mouth, showing that the subject can more clearly recognize human facial expressions.
One of the diagnostic criteria for ASD includes difficulty with eye contact and understanding the direction of gaze. Therefore, changes in gaze patterns serve as an important indicator that the response to social stimuli (others’ expressions) may have improved.
These changes suggest that sensory characteristics unique to ASD, such as “feeling discomfort or stress when making eye contact,” may have been alleviated, demonstrating the anticipated indirect effects on the nervous system via NanoGAS®-FMT.
Future Prospects and Research Collaboration
The purpose of NanoGAS®-FMT is not to achieve short-term fixes, but to balance the nervous system through the intestinal environment and encourage sustainable changes that align with the individual’s unique characteristics. We plan to further strengthen our collaborations with universities and research institutions to continue verifying safety and effectiveness.
The Process of Fecal Microbiota Transplantation for ASD
The general workflow for NanoGAS®-FMT treatment is as follows:
- Initial Consultation and Examination
A doctor reviews current symptoms and daily life to determine eligibility. Subsequently, a general blood test and Intestinal Flora Balance Test are conducted to make a final assessment. ([Link to the flow of Fecal Microbiota Transplantation]) - Preparation of Bacterial Solution and Prior Recording
The bacterial solution used for treatment is manufactured under stringent quality control at a dedicated facility. Before transplantation, we ask you to record daily rhythms, meals, defecation, and behavior in a digital diary (Gut Power-Up Diary) to enable a clear comparison. - Implementation of Fecal Microbiota Transplantation
The frequency of transplantation is determined by the doctor based on age and physical condition, using methods designed to minimize the burden on the patient. - Follow-up After Transplantation
We monitor progress at multiple intervals: 2 weeks, 3 months, 6 months, and 1 year post-transplantation. Doctors and parents share test results and behavioral changes to support the treatment from a long-term perspective.
At NanoGAS®-FMT, we value gradual, sustainable changes in mind and body. Improvements in the intestinal environment are also supported by daily habits such as diet, sleep, and lifestyle rhythms. Therefore, we emphasize a support system that involves the entire family.
Participating Medical Institutions
NanoGAS®-FMT is performed at affiliated medical institutions belonging to The Association for Clinical Research of Fecal Microbiota Transplantation Japan.
- Futamata-kai Medical Corporation Natural Art Clinic (Chiyoda-ku, Tokyo)
With extensive experience in integrated medicine, they have introduced NanoGAS®-FMT as part of a holistic approach to health.
All affiliated institutions use bacterial solutions manufactured at dedicated facilities following a common protocol. If you are considering treatment, please first schedule a consultation to confirm suitability.
[Link: https://fmt.sym-biosis.co.jp/transplantation/partner]
For estimated costs and private practice details, please visit the transplant fee page.
Common Concerns and Responses for Families
Families often ask: “What exactly will change?”, “Will it be painful?”, or “How should we prepare?”NanoGAS®-FMT prioritizes proceeding only after these anxieties are addressed. The transplantation is performed under medical supervision and is neither painful nor overstimulating.
Many families observe that rather than an immediate change, they noticed increased “calmness” after a few months, “softened facial expressions,” and “smoother interactions” at home. We track these changes using both objective data (like Gazefinder) and subjective family records.
Frequently Asked Questions
- Q: How many transplantations are required?
- A: This depends on the individual’s constitution and symptoms. Some feel a significant change after one session, while others improve gradually. The doctor will recommend an optimal plan.
- Q: Are medications or dietary restrictions necessary?
- A: While you should follow your doctor’s advice, maintaining “gut-friendly” habits is highly recommended. [See our Gut Health column: https://fmt.sym-biosis.co.jp/blog]
- Q: Is there an age limit?
- A: NanoGAS®-FMT can be safely administered to children from the age of 2.
Resources and Videos
Explore the mechanisms of the gut-brain axis and intestinal microbiota in our latest columns:
- [The Microbiota-Gut-Brain Axis Explained]
- [Latest Research: ASD and Intestinal Bacteria]
- [Intestinal Environment and Mental Stability]
Watch and Learn on YouTube:
- [FMT Testimonials: Parent Interviews]
- [Relaxed Roundtable with Bacteria (Yurutto Kintachi to Zadankai)]
- [Clinician Interviews]




