Key Differences from Other Fecal Microbiota Transplantations — Seven Features of NanoGAS®-FMT
Procedures and implementation frameworks for Fecal Microbiota Transplantation (FMT) vary by country and medical institution.
Here, we outline the specific procedures and systems adopted by NanoGAS®-FMT from seven perspectives, comparing them to standard methods.
The following descriptions are intended to explain the management processes and equipment systems adopted by The Association for Clinical Research of Fecal Microbiota Transplantation Japan, rather than to imply superiority or inferiority in terms of effectiveness or safety.
1. Antimicrobial-free protocol design
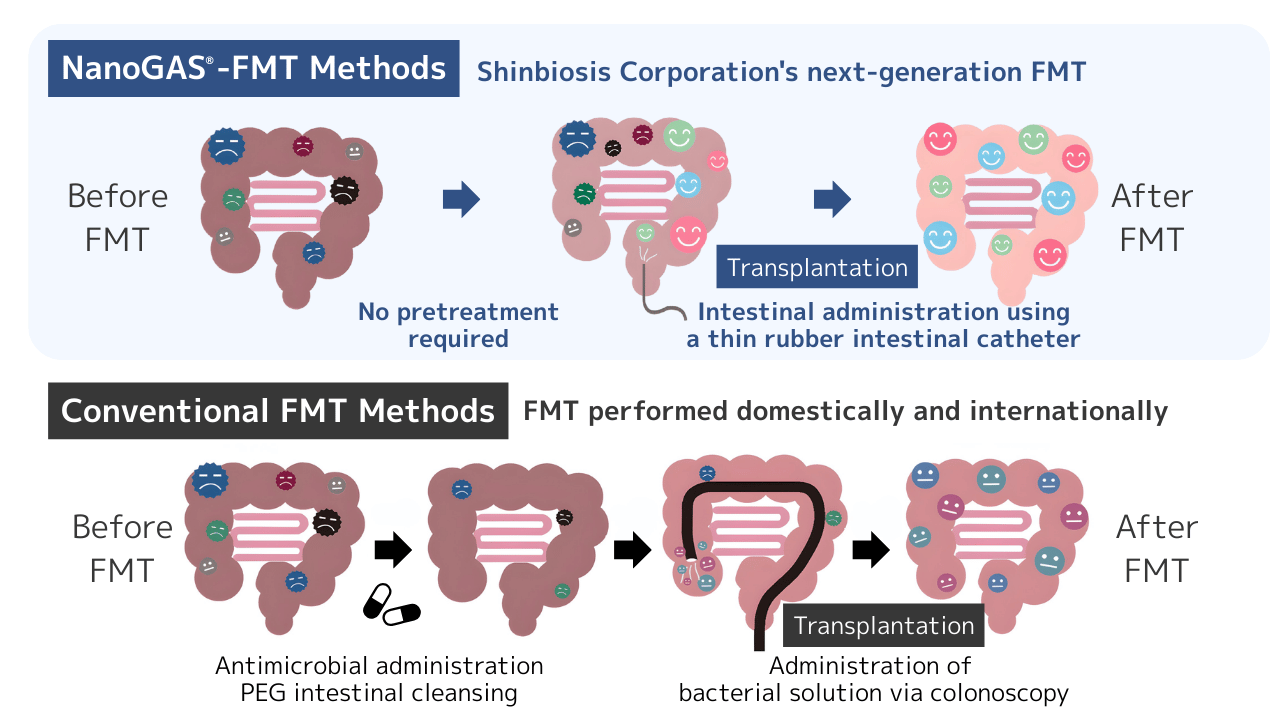
In some conventional FMT cases, medication protocols may be used as a preparatory step before transplantation.
In contrast, NanoGAS®-FMT incorporates a procedure that does not require antimicrobials as a pre-treatment, allowing for the procedure to be performed without significantly disrupting the patient’s existing intestinal environment.
2. Non-endoscopic administration method
It is common for certain FMT procedures to utilize an endoscope for the administration of the bacterial solution.
In NanoGAS®-FMT, we adopt a more minimally invasive administration method using a thin rubber tube.
3. Utilization of proprietary technology for bacterial processing
Since many intestinal bacteria are highly sensitive to oxygen, strict process control is required during manufacturing.
NanoGAS® technology implements rigorous controls to prevent bacterial exposure to oxygen during handling, enabling the production of high-quality bacterial solutions.
4. Comprehensive donor screening and testing protocols
Donors are selected based on rigorous confirmation items regarding health status, infectious disease testing, and lifestyle habits.
In NanoGAS®-FMT, we have established a system to define test items based on domestic and international standards and published guidelines, ensuring appropriate checks are performed before each administration.
5. Manufacturing, storage, and record-keeping in dedicated facilities
From manufacturing and storage to shipping, all processes are conducted within dedicated facilities, with temperature control, hygiene management, and traceability (records) managed for each stage.
By maintaining comprehensive records of all processes, we have established a system where the specific details of each administration can be fully verified at a later date.
6. Research-driven protocol design and clinical considerations
NanoGAS®-FMT designs its process management and testing systems based on the findings of research conducted jointly with domestic medical institutions and universities.
In order to maintain appropriate procedure management as a private-pay treatment, we continue to collect and analyze data in compliance with the Clinical Research Act and related guidelines.
7. A holistic approach to evaluating changes in the microbiome
In NanoGAS®-FMT, we focus on understanding changes in the overall composition of the microbiome rather than the fluctuation of individual bacterial species.
The purpose is not to claim a “therapeutic effect,” but to observe changes in the microbiome over the long term and deepen the understanding of the internal intestinal environment.
NanoGAS®-FMT emphasizes rigorous process management, testing, and the manufacturing environment when working with Fecal Microbiota Transplantation.
On the next page, we will introduce the background behind the design of this method and the philosophy upon which it was founded.
→ To “Characteristics of Our Fecal Microbiota Transplantation“